Today during the intensive coaching of my student under the Final Leap Program, i noticed that many students are not very confident about question on Rate of Reaction (sometimes called Speed of Reaction).
So today, i am going to discuss on this “Must-Score” chapter in O Level Chemistry (same for IB and ICGSE syllabus). Perhaps, i will start a mini-Series on Rate of Reaction for your?quick?revision during this examination period
Different chemical reactions take place at different speeds.
The amount of reactants and products change during a chemical reaction.
We can measure the speed of reaction by measuring:
— how quickly a product is formed per unit time
— how quickly a reactant is used up per unit time
Speed of Reaction = (Change in Amount of Reactant/Product) / time taken
Or (for reaction that produces a Gas),
Speed of Reaction = (Change in Volume of Gas) / time taken
We often see questions in exams relating to RATE GRAPHS – A graph obtained by plotting some measurable quantity (e.g, volume of carbon dioxide produced or loss in mass of flask & contents in the reaction between limestone & hydrochloric acid) against time in order to find the rate of reaction.
Gradient of tangent to any point on the curve
–> Rate of Reaction at any instant
Steeper the gradient
–> faster the reaction
At start of reaction, graph is the steepest
–> Rate at the start of ANY reaction is always Fastest
As reaction progresses, gradient becomes gentler
–> Reaction is Slowing Down
After sometime, the graph levels off
–> point of leveling off signifies the End of Reaction
Let’s check out a question on Rate Graphs!
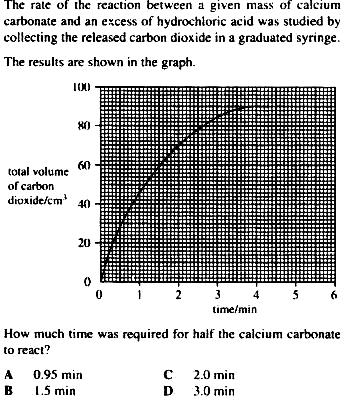
Question 1:
PS: Try it out and remember to leave us your suggested answer in the “Leave A Reply” section below, so that others can come in and dicuss about the answer.