Thought you might find this Redox Reactions question interesting and challenging to solve.
Question by a top secondary school preliminary papers that test you on Oxidation States / Oxidation Numbers.
(Click on Image to have a clearer view)
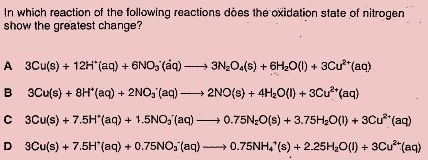
“Whatever you FOCUS on E.X.P.A.N.DS”~ Sean
PS: Check out the “Comments” below for suggested answers.

Hi Gary,
Hope all is well with your revision for Mid-Year exams.
Spot On! Answer is indeed D and your approach towards such Redox question is
correct.
Like what Jennifer has mentioned, the stoichiometry of each species does not
affect the O.S. since the equation is balanced.
Keep up the good work!
To your Academic Success in Chemistry
Sean
Founder & Master Trainer
http://www.SimpleChemConcepts.com
Hi Jennifer,
Very good attempt indeed. The approach you use is correct.
Just note that for (A), the O.S. for N should be +4 instead of +3. Check out the
working again.
Keep up the good work!
To your Academic Success in Chemistry,
Sean
Founder & Master Trainer
http://www.SimpleChemConcepts.com
My answer is D too.But i think the OS for N2 at the products in A should be +4 instead of +3
As Jen said,oxidation state for Nitrogen at the reactants are all +5
A: The OS for N2 at products is +4 so the change of OS is 1.
B: The OS for N2 at products is +2 so the change of OS is 3.
C: The OS for N2 at products is +1 so the change of OS is 4
D:The OS for N2 at products is -3 so the change of OS is 8
Hello.
My answer would be D
For A, B, C, D : initial oxidation state (O.S) of Nitrogen = +5
irregardless of the coefficient of NO3^-1
A: final O.S of Nitrogen = +3
hence, change= +3-(+5) = -2
B: final O.S of Nitrogen = +2
hence, change = +2 – (+5) = -3
C: final O.S of Nitrogen= +1
hence, change = +1 – (+5) = -4
D: final O.S of Nitrogen = -3
hence, change = -3 + – (+5) = -8