Chemical Bonding is an important topic to learn for Chemistry. In fact, fundamentally, it is one of the most important topic that is related to other topics in O-Level Chemistry (and equivalents).
There are 3 types of Bondings:
- Ionic Bonding
- Covalent Bonding
- Metallic Bonding
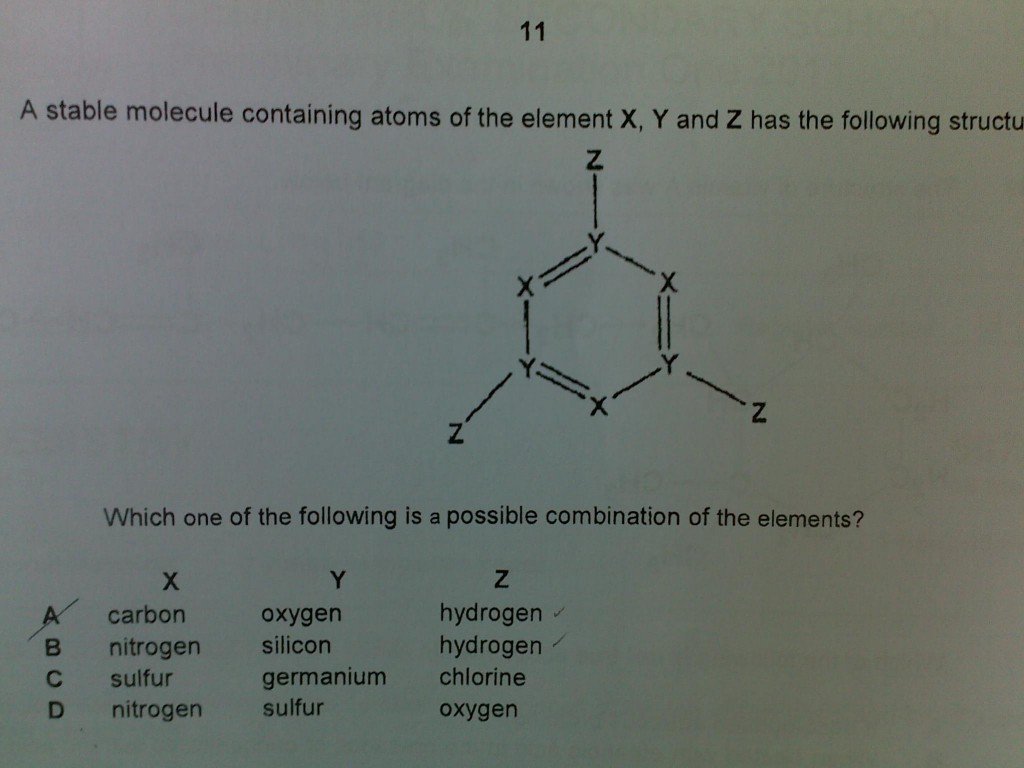
Today, we will discuss a question on bonding that was sent? by a Sec 4 O-Level Chemistry student. I reckon that the question was taken from his school’s preliminary examination paper.
Now, first we need to recognise that the substance is a covalent compound, meaning only covalent bonding exists in the molecule.
To recap, the element that is involved in bonding can have maximum of only 8 valence electrons (exception: Hydrogen with maximum of 2 valence electrons) around itself after bonding.
The 8 electrons can be bonded and can also be unbonded.
Answer is (B) i.e. X = Nitrogen; Y = Silicon and Z = Hydrogen
Reasons as follows:
- Nitrogen is in Group V –> has 5 valence electrons –> 3 electrons used for covalent bonding + 2 unbonded valence electrons
- Silicon is in Group IV –> has 4 valence electrons –> all 4 electrons are used for covalent bonding
- Hydrogen has only 1 valence electron –> electron is used for covalent bonding
Hope you are learning something useful in this post.
PS: If you think this is beneficial to your friend, feel free to forward them this website. =)

ITS VERY USEFUL FOR ALL O/L STUDENTS .I THANK THIS WEBSITE FOR GIVING ME A GOOD KNOWLEDGE ABOUT CHEM THANK U VERY MUCH.